When you read about a promising medical treatment, it often comes from a research study. But the process used in that study looks very different from what happens in a real clinical setting. This gap between research and practice is one of the most important and least discussed topics in modern medicine.
Understanding how treatment protocols are developed, tested, and applied in clinical practice helps you ask better questions. It also helps you make more informed decisions about your health.
What Makes a Research Protocol Different From a Clinical One?
In a research setting, every step of treatment is tightly controlled. Scientists design protocols to answer one specific question: Does this intervention work, and is it safe? They limit variables by selecting patients based on strict criteria, including age, health history, and disease stage.
Clinical practice works differently. A doctor must account for individual health factors, other medications, and practical constraints like access to follow-up care. What works in a controlled lab environment may need significant modification before it is useful to a real patient.
Research protocols are time-bound and structured around data collection, while clinical care is ongoing and responsive. A researcher follows a fixed plan, but a clinician adjusts as new information about the patient becomes available.
This distinction matters for regenerative medicine. Many regenerative approaches, including research into stem cell therapy for conditions like rheumatoid arthritis, type 2 diabetes, and liver disease, are still in active research phases. The protocols used in those studies are not always the same as what a healthcare provider would implement in a clinical setting today.
How Research Protocols Are Designed and Controlled
A clinical research protocol is built around a central hypothesis. Researchers decide in advance how many patients will be enrolled, what the treatment will involve, and how outcomes will be measured. Every variable is documented and standardized.
In Costa Rica, research institutions and regulatory bodies oversee how medical studies are conducted to ensure they meet ethical and scientific standards. This oversight protects participants and ensures findings are valid and trustworthy.
Researchers also document every stage of the study in detail. They track patient responses, side effects, and follow-up results using standardized methods. This process helps evaluate safety, compare outcomes, and identify patterns that may support future clinical use.
Placebo controls, blinding methods, and randomization are all tools used to reduce bias. A participant in a stem cell study may not know whether they received an active treatment or a control intervention. This level of control is not practical in clinical care, where the goal is to treat the individual rather than generate data.
How Clinical Protocols Are Adapted for Real-World Use
Clinical protocols are designed to be practical. They account for the fact that patients have different needs, different access to care, and different responses to treatment. A research trial for COPD or spinal cord injury recovery may have required weekly visits, but clinical practice might restructure that into monthly check-ins with remote monitoring.
When moving from research to practice, providers also consider local infrastructure and available resources. A protocol developed in a large research center may need significant adaptation before it works in a community clinic or wellness setting in Costa Rica.
Physicians and specialists review published evidence and adapt it based on local resources and patient populations. The goal is not to replicate the research study exactly, but to apply what worked in research to an approach that works in real life.
For conditions like osteoarthritis, chronic pain, or autoimmune diseases such as lupus or psoriasis, clinical protocols often combine multiple approaches. Regenerative strategies may work alongside physical therapy, nutritional support, and standard medical care. A patient’s condition, overall health, and recovery goals all help shape the final treatment plan.
Why the Gap Between Research and Practice Exists
The time between a promising research finding and its routine clinical use is often longer than people expect. Studies estimate it can take a decade or more for research evidence to consistently shape clinical behavior.
Regulatory review is one reason. Before a treatment can be offered clinically, it must meet the standards set by health authorities. In Costa Rica, this means demonstrating both safety and efficacy through a defined evidence base.
Generalizability is another factor. A stem cell study for heart tissue repair may focus only on patients within a specific age group or with a certain type of heart damage. Applying those findings to a broader population requires more research, clinical judgment, and proper provider training.
What This Means for Patients Exploring Regenerative Options
If you are exploring regenerative medicine for conditions like alopecia, multiple sclerosis, or Crohn’s, knowing the difference between what is being studied and what is clinically available will help you evaluate your options clearly. Research findings are valuable, but they represent the beginning of a process, not the end.
A study showing reduced inflammation in a controlled setting is meaningful, but it tells you less about what a clinical provider would do with that information in practice. It is worth asking whether an approach is based on clinical evidence or is still in active research, and how it fits alongside your existing care.
Knowing that a protocol is still being studied does not mean it lacks merit. It means the process of validating and refining it is still ongoing.
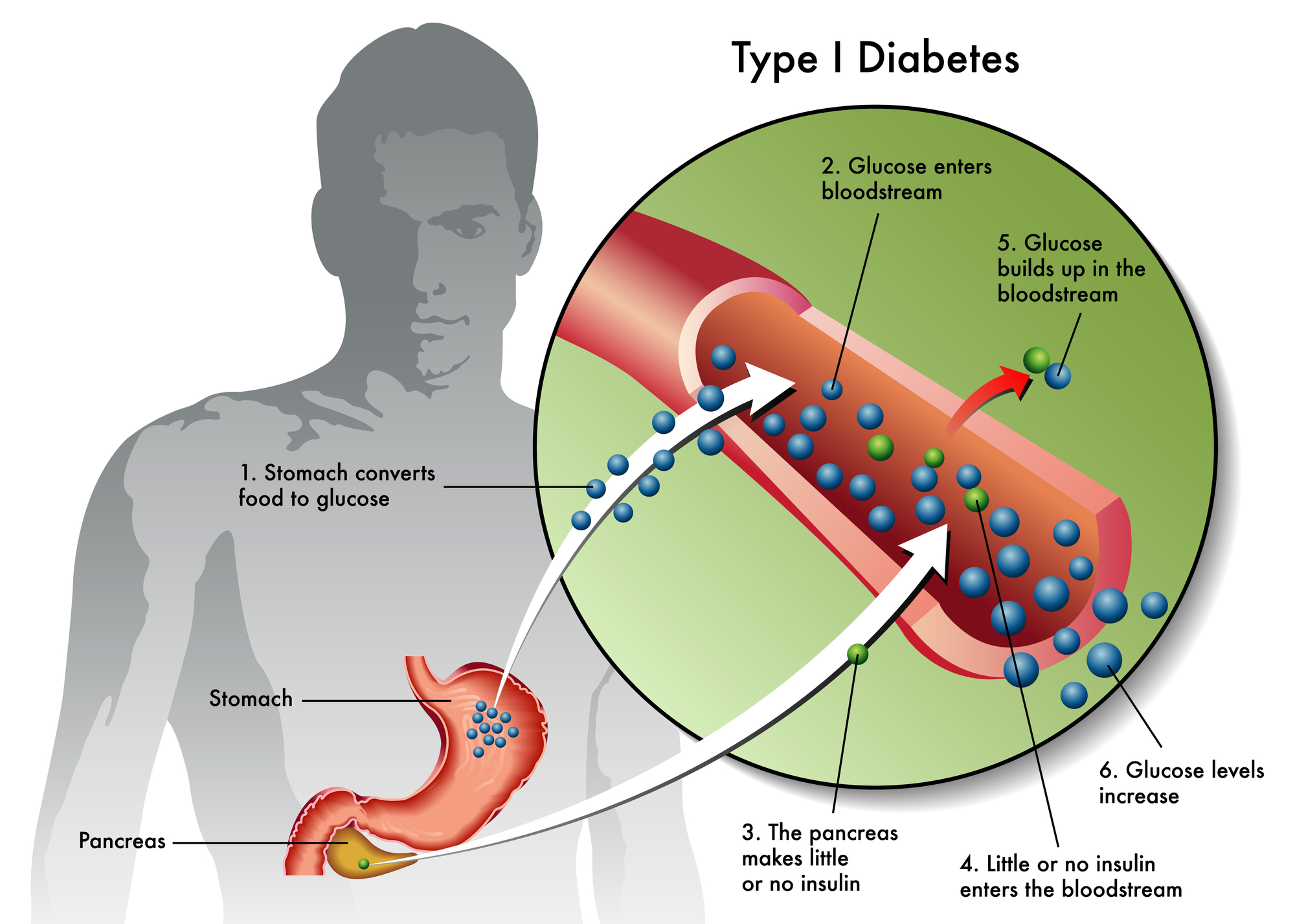
Regenerative medicine continues to advance, and research into conditions like diabetes type 1 diabetes, COVID-19 recovery, and inflammatory diseases is producing new knowledge regularly. Staying informed means understanding both what the research shows and where it stands in the process of becoming clinical practice.
Take the Next Step With Cellebration Wellness
Understanding how treatment protocols differ between research and clinical settings is an important part of exploring regenerative care. Knowing where a therapy stands in its development can help you have more informed conversations with medical professionals about what may be appropriate for your condition.
Cellebration Wellness focuses on education and wellness approaches inspired by the latest advances in regenerative science. Speaking with a qualified professional can help you better understand available options and decide what may fit your needs.
Contact Cellebration Wellness today at (858) 258-5090 to learn more about your regenerative health options.